Climate conference in Glasgow
- Details
We will fight the climate crisis internationally or we will fail // German debate more European and European debate more international // Important approaches in the proposal on European emissions trading
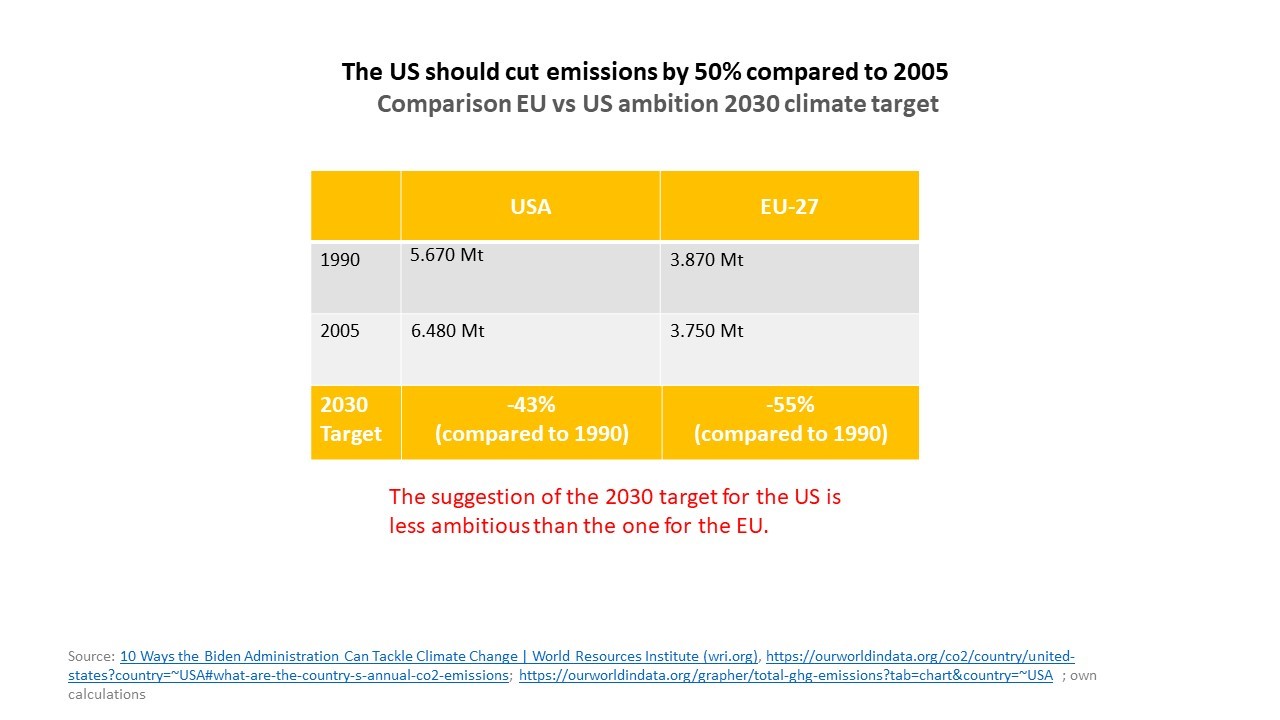
"We will fight the climate catastrophe internationally or not at all," declared the environmental policy spokesman of the largest group in the European Parliament (EPP, Christian Democrats), MEP Dr. Peter Liese, with regards to the climate conference in Glasgow starting on Sunday. “Unfortunately, neither climate-damaging greenhouse gases nor their effects stop at the border. That's why we do not have to discuss first and foremost whether the European climate targets need to be raised even further, but how we can get the rest of the world on board. There are different assessments of how ambitious the European Union's climate goal of reducing emissions by 55% by 2030 is. In my view, it is much more ambitious than the target that President Biden has set for the US.
European Commission's climate package can be the BioNTech vaccination against high energy prices
- Details
Peter Liese on the discussion on energy prices in the European Union: Relief inevitable in the short term, invest in renewable energies and energy efficiency in the medium and long term to be immune against high prices of fossil fuels
"The European Commission's proposals on the climate and energy package (Fit for 55) can be the BioNTech/Pfizer vaccination against high energy prices," said the spokesman on health and environment of the largest group in the European Parliament (EPP, Christian Democrats), Dr. med. Peter Liese, in view of the discussion on high energy prices at the European Council on Wednesday and Thursday. "We must take serious that many people are being hit hard by the rise in energy prices and short-term aid is unavoidable. But we should not shelve the medium and long-term climate change agenda.
FDA recommends emergency approval for BioNTech Pfizer vaccine for 5-11 year old children
- Details
Conditional marketing authorisation in Europe gives more certainty, nevertheless the company and EMA must hurry now // Important especially for children with pre-existing conditions // Off-label use also justified in very important cases
After an advisory panel of the US Food and Drug Administration (FDA) recommended an emergency approval for the vaccine of BioNTech/Pfizer for five to eleven year old children tonight, the health policy spokesman of the largest group in the European Parliament (EPP-Christian Democrats), MEP Peter Liese, urges a careful, but speedy review of the application for approval also at the European Medicines Agency. This is particularly important for children with pre-existing conditions. The EU's delay compared to the US is not only due to the European Medicines Agency, which Liese called on to work more quickly without compromising safety, but also to the company, which still has to submit additional data. "Unlike the US, there is a somewhat more extensive review process in the EU. The conditional marketing authorisation involves the examination of additional data on effects and side effects and more experts analyse the data. In addition, unlike in the US, the manufacturer is liable in the EU procedure. Especially in the case of children, it is important to ensure the greatest possible safety. Nevertheless, the procedure must not take too long now. A delay of a few weeks compared to the US can be justified by the additional safety, but the authorisation must definitely be granted this year if no serious problems are found," said the doctor and MEP.
Read more: FDA recommends emergency approval for BioNTech Pfizer vaccine for 5-11 year old children
Warning of shortages in diagnostic medical devices - European Commission proposes extended transition periods for in vitro diagnostic medical devices
- Details
Right and necessary step - We must ensure that patients, hospitals and research institutions remain equipped with necessary diagnostic tests
The European Commission today proposed extended transition periods for certain in vitro medical devices to avoid supply shortages. Already in 2017, the EU institutions agreed on a new regulation on in vitro diagnostic medical devices, which will enter into force in May 2022. This regulates market access for all medical devices for medical laboratory testing of samples derived from the human body (IVDs), such as HIV tests, pregnancy tests or corona tests. The health policy spokesperson of the largest group in the European Parliament (EPP-Christian Democrats), Dr. Peter Liese, welcomed the proposal. "Already before the summer break, together with colleagues from all democratic groups in the European Parliament, I wrote to the Commission in order to find a solution to any bottlenecks.The Regulation remains right, necessary and in force. The extended transition periods proposed today will ensure that patients, hospitals, research facilities and other medical institutions will continue to be equipped with all the necessary diagnostic tests after the new regulation enters into force in May 2022," said the doctor and MEP.